
Home / News
-
-

-
Paige, a leader in next-generation AI technology, and the Breast International Group (BIG), a leading international non-profit organization dedicated to breast cancer research, today announced a new partnership to conduct collaborative research aimed at improving breast cancer diagnosis, treatment, and patient outcomes.2025-06-11View More
-
-
-

-
Quest Diagnostics (NYSE: DGX), a leading provider of diagnostic information services, today announced a collaboration with The University of Texas MD Anderson Cancer Center (MD Anderson) designed to improve the assessment of elevated risk of cancer in individuals who would benefit from medically appropriate cancer screenings.2025-06-11View More
-
-
-

-
Perspectives on the IVD Market in China 2025 We are pleased to invite you to our upcoming webinar, Perspectives on the IVD Market in China 2025, tailored for overseas IVD manufacturers seeking to better ..2025-06-10View More
-
-
-

-
As metabolic dysfunction-associated steatohepatitis (MASH) emerges as one of the most dangerous and underdiagnosed drivers of liver failure―affecting more than 250 million people worldwide―Echosens, the leader in non-invasive liver diagnostics, and Boehringer Ingelheim, a global biopharmaceutical company, today announced an expansion of their long-standing partnership to change the trajectory of the disease by moving beyond clinical trials to focus on early detection, diagnosis, and access to care.2025-06-10View More
-
-
-

-
Diasorin (FTSE MIB: DIA) today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the LIAISON PLEX® Gram-Positive Blood Culture Assay, the final syndromic blood culture panel for the microbiological diagnosis of bloodstream infections on the LIAISON PLEX®.2025-06-10View More
-
-
-

-
2. Dry Chemical Technology The biggest difference between “dry chemistry” and traditional “wet chemistry” (i.e., solution chemistry) is the media involved in the chemical reactions. In this method, the liqui..2025-06-10View More
-
-
-

-
Thermo Fisher and Biomobio Sign Strategic Agreement in Spatial Imaging Thermo Fisher Scientific and Biomobio have successfully concluded a strategic cooperation agreement for the EVOS S1000..2025-06-09View More
-
-
-

-
New data published in JAMA Internal Medicine on Friday from MD Anderson Cancer Center researchers found that mail-in self-collection kits for human papillomavirus testing more than doubled cervical cancer screening participation among never- and under-screened patients.2025-06-09View More
-
-
-

-
Burning Rock Biotech Limited (NASDAQ: BNR, the “Company” or “Burning Rock”), a company focused on the application of next generation sequencing (NGS) technology in the field of precision oncology, today reported financial results for the three months ended March 31, 2025.2025-06-09View More
-
-
-

-
Fujirebio, a leading innovator in in-vitro diagnostics, today announced a collaboration with Stanford Medicine (Location: Palo Alto, California, USA) to advance research and innovation in the field of infectious disease testing. This collaboration aims to accelerate the adoption of ultrasensitive immunoassays that incorporate single-molecule counting technology developed by Fujirebio’s Silicon Valley wholly-owned subsidiary, Fluxus, Inc. Greater test sensitivity can better inform treatment decisions in the clinic, as well as accelerate studies towards therapeutics and preventive strategies against infectious disease threats worldwide.2025-06-09View More
-
-
-

-
Randox Laboratories announced that the U.S. Food and Drug Administration (FDA) has approved the De Novo application for the company’s first Companion Diagnostic (CDx). This is a major achievement for Randox following over 3 years of innovative development and collaboration with Novo Nordisk. The Companion Diagnostic was developed as a new option for determining the amount of medication a patient with hemophilia has received.2025-06-09View More
-
-
-

-
Cepheid today announced that Health Canada has issued Cepheid a medical device licence for Xpert® HIV-1 Viral Load XC, a next-generation extended-coverage (XC) test intended to aid in assessing HIV viral load levels, which are used to monitor effectiveness of antiretroviral treatment.2025-06-08View More
-
-
-
-
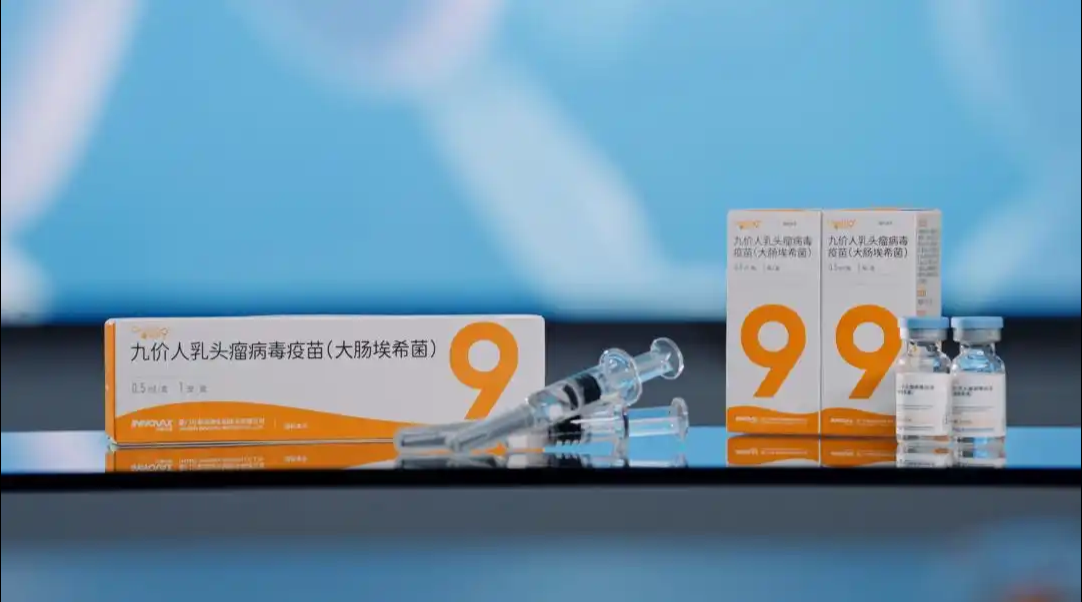
China's drug regulator has approved the country's first domestically developed 9-valent human papillomavirus (HPV) vaccine, ending over a decade of foreign dominance in the market.2025-06-08View More
-
-
-

-
Technical Platform POCT technology has gradually evolved from classical immunochromatography to more sensitive platforms hosting multiple detection technologies, such as luminescence and molecular detection. ..2025-06-04View More
-
-
-

-
Guardant Health, Inc. (Nasdaq: GH), a leading precision oncology company, today announced that its Shield multi-cancer detection (MCD) test has been granted Breakthrough Device designation from the U.S. Food and Drug Administration (FDA). The Shield MCD test is a methylation-based blood test for the multi-cancer screening of multiple cancer types including bladder, colorectal, esophageal, gastric, liver, lung, ovarian and pancreas cancer in individuals aged 45 or older who are at typical average risk for cancer.2025-06-04View More
-
-
-

-
QuidelOrtho Corporation (Nasdaq: QDEL) (the "Company" or “QuidelOrtho”), a global provider of innovative in vitro diagnostic technologies designed for point-of-care settings, clinical labs and transfusion medicine, today announced a refocusing of its molecular diagnostics strategy. This strategy includes the intent to acquire full ownership of LEX Diagnostics after 510(k) clearance by the U.S. Food and Drug Administration ("FDA") for consideration at closing of approximately $100 million and plans to discontinue the Company's Savanna platform development.2025-06-04View More
-
-
-

-
PathAI, a leading provider of AI-powered pathology solutions, and Northwestern Medicine, Chicago’s premier integrated academic health system, today announced a strategic collaboration to transform pathology diagnostics through artificial intelligence. This multi-year collaboration will see Northwestern Medicine implement PathAI’s AISight digital pathology image management system with the goal of driving efficiency and quality in pathology operations. In addition to the digital innovation efforts, this joint effort establishes a broad framework for joint research initiatives, clinical innovation programs, and co-development of new AI-powered diagnostic tools aimed at improving patient outcomes.2025-06-03View More
-
-
-

-
QIAGEN (NYSE: QGEN; Frankfurt Prime Standard: QIA) today announced the expansion of its oncology diagnostics portfolio with two strategic partnerships to advance the use of minimal residual disease (MRD) testing in clinical trials to support pharma co-development projects for companion diagnostics. The new collaborations with Tracer Biotechnologies and Foresight Diagnostics expand QIAGEN’s reach in MRD testing and cover solid tumors and hematological cancers.2025-06-03View More
-
-
-

-
From May 21-23, 2025, Lei Haichao, Director of the National Health Commission of the P.R.C, led a delegation on an official visit to France.2025-05-30View More
-
-
-

-
Introduction Point-of-care testing (POCT) is a subfield of in vitro diagnostics (IVD), where portable analytical instruments and supporting reagents are used to obtain test results quickly at the sampling site. P..2025-05-30View More
-

- CAIVD WeChat
Subscription Account

- CAIVD WeChat
Channels
China Association of In-vitro Diagnostics
Part of the information in our website is from the internet.
If by any chance it violates your rights, please contact us.

